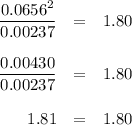Phosphorus pentachloride decomposes according to the chemical equation pcl5(g)↽−−⇀pcl3(g)+cl2(=1.80 at 250 ∘c a 0.197 mol sample of pcl5(g) is injected into an empty 2.90 l reaction vessel held at 250 ∘c. calculate the concentrations of pcl5(g) and pcl3(g) at equilibrium.

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 19:00, qwerty8364
Asyringe contains 56.05 ml of gas at 315.1 k. what volume will that gas occupy if the temperature is increased to 380.5 k? a) 12.41 b) 46.42 c) 67.68 d) 81.74
Answers: 1

Chemistry, 22.06.2019 03:00, ian2006huang
Which of these would be caused by a chemical change? a) the formation of lava. b) sedimantary rock layering over time. c) metamorphic rock forming from igneous. d) metamorphic rock eroding to form sedimentary rock.
Answers: 3

Chemistry, 22.06.2019 09:20, UsedForSchool2018
Which of these statements explains the difference between nuclear binding energy and the strong nuclear force ?
Answers: 3

Chemistry, 22.06.2019 20:30, allofthosefruit
Draw a line graph showing the relationship between temperature in kelvin as a function of kinetic energy.
Answers: 3
You know the right answer?
Phosphorus pentachloride decomposes according to the chemical equation pcl5(g)↽−−⇀pcl3(g)+cl2(=1.80...
Questions in other subjects:

Arts, 25.09.2019 23:00


History, 25.09.2019 23:00




Chemistry, 25.09.2019 23:00

History, 25.09.2019 23:00

Social Studies, 25.09.2019 23:00

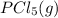 is 0.0655 M and
is 0.0655 M and  is 0.00240 M at equilibrium.
is 0.00240 M at equilibrium.


![[PCl_3]=x](/tpl/images/0128/9812/3c44a.png)
![[Cl_2] = x](/tpl/images/0128/9812/a7a23.png)
![=[PCl_5]= (0.0697- x)](/tpl/images/0128/9812/ead0f.png)
![K_c=\frac{[PCl_3][Cl_2]}{[PCl_5]}\\\\1.80=\frac{x\times x}{(0.0679-x)}\\\\x = 0.0655](/tpl/images/0128/9812/79ef0.png)
![=[PCl_5]= (0.0679- x) = (0.0679 -0.0655 )M=0.00240 M](/tpl/images/0128/9812/ac2f8.png)
![= [Cl_2] = x = 0.0655 M](/tpl/images/0128/9812/6faf5.png)
![\text{[PCl$_{5}$]} = \dfrac{\text{0.197 mol}}{\text{2.90 L}} = \text{0.067 93 mol/L}\\\\](/tpl/images/0128/9812/66356.png)
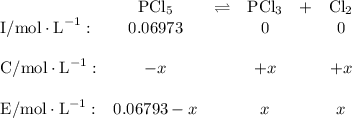
![K_{\text{c}} = \dfrac{\text{[PCl$_3$][Cl$_2$]}}{\text{[PCl$_5$]}} = \dfrac{x^{2}}{0.06793-x} = 1.80\\\\\begin{array}{rcl}\\x^{2}& = & 1.80(0.06793 - x)\\x^{2& = & 0.1223 - 1.80x\\x^{2} + 1.80x - 0.1223& = & 0\\x & = & \mathbf{0.0656}\\\end{array}](/tpl/images/0128/9812/cf1bb.png)