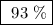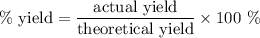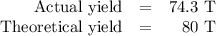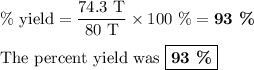Chemistry, 22.07.2019 05:10 meganwintergirl
Achemical factory is making soda ash (na2co3) from sodium bicarbonate. the production manager calculates they will make 80 tons of soda ash in the next 8 hour shift. at the end of the shift they weigh the produced soda ash at 74.3 tons. what was their percent yield?

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 09:00, hellodarkness14
This chart lists four kinds of polymers and their sources. what can be known about all four polymers, despite their differences? they come from living things. they share ionic carbon bonds. they are at least 100 monomers long. they are made of repeating subunits.
Answers: 3


Chemistry, 22.06.2019 16:30, ddmoorehouseov75lc
Correct relationship between molecular formula and empirical formula
Answers: 1

Chemistry, 22.06.2019 21:20, paatnguyyen
One way in which the useful metal copper is produced is by dissolving the mineral azurite, which contains copper(ii) carbonate, in concentrated sulfuric acid. the sulfuric acid reacts with the copper(ii) carbonate to produce a blue solution of copper(ii) sulfate. scrap iron is then added to this solution, and pure copper metal precipitates out because of the following chemical reaction: (s) (aq) (s) (aq) suppose an industrial quality-control chemist analyzes a sample from a copper processing plant in the following way. he adds powdered iron to a copper(ii) sulfate sample from the plant until no more copper will precipitate. he then washes, dries, and weighs the precipitate, and finds that it has a mass of .
Answers: 2
You know the right answer?
Achemical factory is making soda ash (na2co3) from sodium bicarbonate. the production manager calcul...
Questions in other subjects:


Mathematics, 25.09.2020 01:01


Mathematics, 25.09.2020 01:01


Mathematics, 25.09.2020 01:01

Mathematics, 25.09.2020 01:01



Mathematics, 25.09.2020 01:01