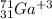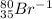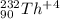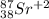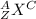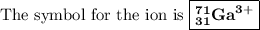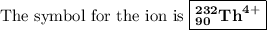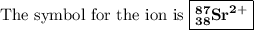Chemistry, 20.07.2019 06:10 brookcoyanpeus5y
Write the symbol for each of the following ions: (a) the ion with a 3+ charge, 28 electrons, and a mass number of 71 (b) the ion with 36 electrons, 35 protons, and 45 neutrons (c) the ion with 86 electrons, 142 neutrons, and a 4+ charge (d) the ion with a 2+ charge, atomic number 38, and mass number 87

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 02:00, natalie857123
For each of the following types of reactions, write a general reaction formula in the symbolic form—for example, a + b → ab. single-displacement double-displacement synthesis decomposition
Answers: 1

Chemistry, 22.06.2019 08:00, PrincessKeliah5538
Me i dont know what to do! the table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 10 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of the two atoms? both are unreactive. both are highly reactive. a is unreactive and d is reactive. a is reactive and d is unreactive.
Answers: 1

Chemistry, 22.06.2019 08:00, gomezyonathan93
An observation that requires measurement is called quantitative observable or qualitative
Answers: 1
You know the right answer?
Write the symbol for each of the following ions: (a) the ion with a 3+ charge, 28 electrons, and a...
Questions in other subjects:


English, 27.07.2019 03:00


English, 27.07.2019 03:00




History, 27.07.2019 03:00


Social Studies, 27.07.2019 03:00