
Chemistry, 18.07.2019 23:30 kalialee2424
How many grams of nahco3 (fm 84.01 g/mol) should be mixed with na2co3 to produce a 1.00 l buffer solution with ph 9.50. the final concentration of na2co3 in this solution is 0.10 m. pka1 = 6.37 and pka2 = 10.33 for h2co3.

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 15:00, emmalie52
Phosphorous acid, h3po3(aq) , is a diprotic oxyacid that is an important compound in industry and agriculture. p k a1 p k a2 1.30 6.70 calculate the ph for each of the points in the titration of 50.0 ml of 1.5 m h3po3(aq) 1.5 m h 3 po 3 ( aq ) with 1.5 m koh(aq). 1.5 m koh ( aq ) .
Answers: 1


Chemistry, 22.06.2019 22:30, xlebrny7831
Amedication is given at a dosage of 3.000 mg of medication per kg of body weight. if 0.1500 g of medication is given, then what was the patient's weight in pounds (lbs)? there are 453.59g in 1 lb.
Answers: 2

Chemistry, 23.06.2019 02:00, ItzAquaZ1449
Alice did an experiment to find the relationship between the angle at which a ray of light strikes a mirror and the angle at which the mirror reflects the light. she placed a ray box in front of a mirror. she changed the angle at which the light from the ray box struck the mirror and noted the corresponding angle at which the mirror reflected the light. which of the following is the dependent variable in this experiment? the mirror used to reflect the light the ray box used as the source of light angle at which the light from the ray box strikes the mirror angle at which the mirror reflects the light from the ray box
Answers: 2
You know the right answer?
How many grams of nahco3 (fm 84.01 g/mol) should be mixed with na2co3 to produce a 1.00 l buffer sol...
Questions in other subjects:


Mathematics, 30.01.2020 22:46





English, 30.01.2020 22:46

Mathematics, 30.01.2020 22:46

Computers and Technology, 30.01.2020 22:46

Mathematics, 30.01.2020 22:46

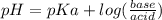
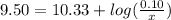
 is the concentration of sodium bicarbonate)
is the concentration of sodium bicarbonate)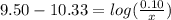
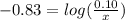
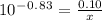
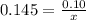
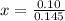
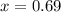
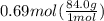
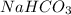 will be required.
will be required. 

