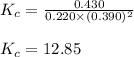Chemistry, 09.07.2019 04:30 dakotalynnwillis01
Amixture initially contains a, b, and c in the following concentrations: [a] = 0.350 m , [b] = 0.650 m , and [c] = 0.300 m . the following reaction occurs and equilibrium is established: a+2b⇌c at equilibrium, [a] = 0.220 m and [c] = 0.430 m . calculate the value of the equilibrium constant, kc.

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 01:30, Slycooper5959
Idon't really understand this can you me and show your work.☺☺[ chemistry b] subject [ electron transfer in lonic bonds]grade( 12)
Answers: 1

Chemistry, 22.06.2019 11:00, 21villalobosjabez
Which type of fossil does this image depict?
Answers: 1

Chemistry, 22.06.2019 19:20, Lovelybunny321
15. which of the following is not human-caused groundwater pollution? a. water in an aquifer dissolves elements such as arsenic and mercury from surrounding rock. b. water in an aquifer is contaminated by leachate that seeps into the ground from a landfill. c. water in an aquifer becomes polluted with chemicals used in hydraulic fracturing, or fracking. d. water in an aquifer absorbs harmful bacteria from the drainage field of a septic tank.
Answers: 1

Chemistry, 22.06.2019 20:30, Schoolworkspace453
Consider the following unbalanced equation for the combustion of hexane: αc6h14(g)+βo2(g)→γco2(g)+δh2o(g) part a balance the equation. give your answer as an ordered set of numbers α, β, γ, use the least possible integers for the coefficients. α α , β, γ, δ = nothing request answer part b determine how many moles of o2 are required to react completely with 5.6 moles c6h14. express your answer using two significant figures. n n = nothing mol request answer provide feedback
Answers: 2
You know the right answer?
Amixture initially contains a, b, and c in the following concentrations: [a] = 0.350 m , [b] = 0.65...
Questions in other subjects:










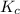 for the given reaction is 12.85.
for the given reaction is 12.85.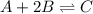
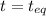 (0.350 - x) (0.650 - 2x) (0.300 + x)
(0.350 - x) (0.650 - 2x) (0.300 + x)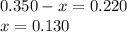
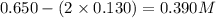
![K_c=\frac{[C]}{[A][B]^2}](/tpl/images/0068/1022/240ef.png)