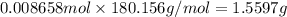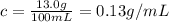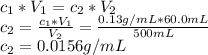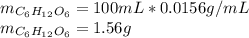Chemistry, 09.07.2019 02:10 paulasuarez8276
Astudent placed 13.0 g of glucose (c6h12o6) in a volumetric flask, added enough water to dissolve the glucose by swirling, then carefully added additional water until the 100.-ml mark on the neck of the flask was reached. the flask was then shaken until the solution was uniform. a 60.0-ml sample of this glucose solution was diluted to 0.500 l. how many grams of glucose are in 100. ml of the final solution?

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 15:40, lexybellx3
Achemistry student weighs out of phosphoric acid , a triprotic acid, into a volumetric flask and dilutes to the mark with distilled water. he plans to titrate the acid with solution. calculate the volume of solution the student will need to add to reach the final equivalence point. round your answer to significant digits.
Answers: 3


Chemistry, 22.06.2019 07:30, SchoolFirst9811
The scheme below is from a series of reactions that are part of a synthesis of vitamin a. answer the following questions with reference to this scheme. (i) what is "reagent a"? (ii) draw a step-by-step mechanism which explains the formation of compound c from compound b (iii) which reagents would you use to form compound e from compounds c and d (reagents b and c)? for each reagent suggested above in (ii) explain the role of the reagent in the reaction to (iv) form compound e. you may wish to do this by drawing a mechanism. 1. addition of reagent a но reagent a 2. н, о" thо oh нон-с compound a. compound b. compound c .ch-оh 1. reagent b "сно 2. reagent c сh oh compound e. compound d.
Answers: 2
You know the right answer?
Astudent placed 13.0 g of glucose (c6h12o6) in a volumetric flask, added enough water to dissolve th...
Questions in other subjects:


Mathematics, 18.04.2020 06:18


English, 18.04.2020 06:18



History, 18.04.2020 06:18

Geography, 18.04.2020 06:18


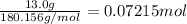
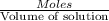
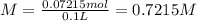
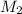
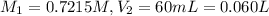
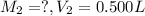
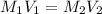 (Dilution)
(Dilution)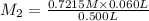
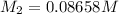
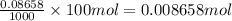 of glucose
of glucose