
Chemistry, 05.02.2020 12:00 hillisaiah734
The reform reaction between steam and gaseous methane (ch4) produces "synthesis gas," a mixture of carbon monoxide gas and dihydrogen gas. synthesis gas is one of the most widely used industrial chemicals, and is the major industrial source of hydrogen.
suppose a chemical engineer studying a new catalyst for the reform reaction finds that 924. liters per second of methane are consumed when the reaction is run at 261.°c and 0.96atm. calculate the rate at which dihydrogen is being produced. give your answer in kilograms per second. round your answer to 2 significant digits.

Answers: 3


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 05:50, aylengarcia090
What are transitions between a liquid and gas called? identify which way they are transitioning
Answers: 2

Chemistry, 22.06.2019 08:30, Blaise2653
Joan writes four numbers on the board in standard form, and then she writes their scientific notation
Answers: 1

Chemistry, 22.06.2019 19:00, nayashuntel
How many liters of ethylene glycol antifreeze (c2h6o2), with a density of 1.100 g/l, would you add to your car radiator containing 15.0 kg of water if you needed to protect your engine to - 21.5°c? for water, kf = 1.86°c m -1.
Answers: 1
You know the right answer?
The reform reaction between steam and gaseous methane (ch4) produces "synthesis gas," a mixture of c...
Questions in other subjects:




Mathematics, 18.09.2019 03:30


History, 18.09.2019 03:30




Mathematics, 18.09.2019 03:30

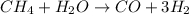 Haber reaction
Haber reaction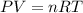
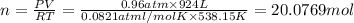
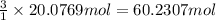 of dihydrogen
of dihydrogen


