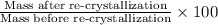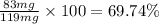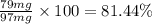Chemistry, 06.07.2019 03:20 savannahvargas512
Amixture of two compounds, a and b, was separated by extraction. after the compounds were dried, their masses were found to be: 119 mg of compound a and 97 mg of compound b. both compounds were recrystallized and weighed again. after recrystallization, the mass of compound a was 83 mg and the mass of compound b was 79 mg. calculate the percent recovery from recrystallization for both compounds.

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 03:40, 19thomasar
Kc = 0.040 for the system below at 450oc. if a reaction is initiated with 0.40 mole of cl2 and 0.40 mole of pcl3 in a 2.0 liter container, what is the equilibrium concentration of cl2 in the same system? pcl5(g) ⇄ pcl3(g) + cl2(g)
Answers: 3

Chemistry, 22.06.2019 15:00, kandi2565
Large helium-filled balloons are used to lift scientific equipment to high altitudes. what is the pressure inside such a balloon if it starts out at sea level with a temperature of 10.0ºc and rises to an altitude where its volume is twenty times the original volume and its temperature is – 50.0ºc ?
Answers: 2


Chemistry, 22.06.2019 16:30, Eddie997
For the reaction shown, calculate how many moles of no2 form when each of the following completely reacts. 2n2o5(g)→4no2(g)+o2(g) part a 1.0 mol n2o5 express your answer using two significant figures. nothing mol m o l request answer part b 5.4 mol n2o5 express your answer using two significant figures.
Answers: 2
You know the right answer?
Amixture of two compounds, a and b, was separated by extraction. after the compounds were dried, the...
Questions in other subjects:





Mathematics, 24.05.2020 07:58

English, 24.05.2020 07:58

Mathematics, 24.05.2020 07:59


Mathematics, 24.05.2020 07:59