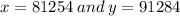Chemistry, 06.07.2019 02:30 coopyishome
Iron (fe) undergoes an allotropic transformation at 912°c: upon heating from a bcc (α phase) to an fcc (γ phase). accompanying this transformation is a change in the atomic radius of fe—from rbcc = 0.12584 nm to rfcc = 0.12894 nm—and, in addition, a change in density (and volume). compute the percentage volume change associated with this reaction. indicate a decreasing volume by a negative number.

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 12:10, coastieltp58aeg
Building glycogen from glucose molecules is an example of
Answers: 3

Chemistry, 22.06.2019 13:30, annanikherrera
If the concentration of phosphate in the cytosol is 2.0 mm and the concentration of phosphate in the surrounding fluid is 0.1 mm, how could the cell increase the concentration of phosphate in the cytosol? a) passive transportb) diffusionc) active transportd) osmosise) facilitated diffusion
Answers: 3


Chemistry, 23.06.2019 02:00, FailingstudentXD
Calculate the molarity of each aqueous solution: a. 78.0 ml of 0.240 m naoh diluted to 0.250 l with water b. 38.5 ml of 1.2 m hno3 diluted to 0.130 l with water
Answers: 1
You know the right answer?
Iron (fe) undergoes an allotropic transformation at 912°c: upon heating from a bcc (α phase) to an...
Questions in other subjects:



Mathematics, 20.02.2021 08:20





Mathematics, 20.02.2021 08:20


English, 20.02.2021 08:20