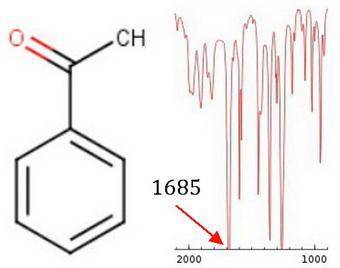
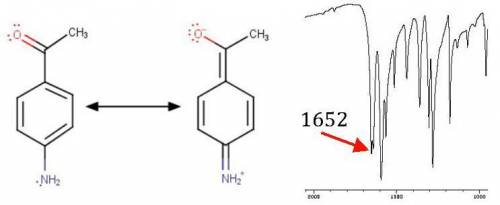
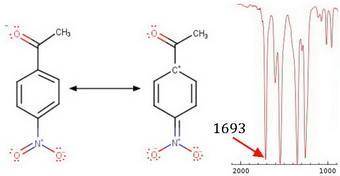
Substitution of an amino group on the para position of acetophenone shifts the cjo frequency from about 1685 to 1652 cm−1 , whereas a nitro group attached to the para position yields a cjo frequency of 1693 cm−1 . explain the shift for each substituent from the 1685 cm−1 base value for acetophenone

Answers: 2


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 03:00, parisaidan366
What happened in 2012 and how does it illustrate the importance of understanding the sun and how it works?
Answers: 3

Chemistry, 22.06.2019 09:00, tbiles99
What type of energy do chemical bonds have? what type of energy is it converted to during chemical reactions? question 15 options: chemical bonds have kinetic energy, which is converted to potential energy during chemical reactions. chemical bonds have electric energy, which is converted to potential energy during chemical reactions. chemical bonds have heat energy, which is converted to kinetic energy during chemical reactions. chemical bonds have potential energy, which is converted to heat energy during chemical reactions.
Answers: 1

Chemistry, 22.06.2019 12:30, emmalybrown
A50.0 ml sample of gas at 20.0 atm of pressure is compressed to 40.0 atm of pressure at constant temperature. what is the new volume? 0.0100 ml 0.325 ml 25.0 ml 100. ml
Answers: 1
You know the right answer?
Substitution of an amino group on the para position of acetophenone shifts the cjo frequency from ab...
Questions in other subjects:


English, 26.04.2020 00:51

Mathematics, 26.04.2020 00:51

Mathematics, 26.04.2020 00:51


Mathematics, 26.04.2020 00:51


Geography, 26.04.2020 00:51

Social Studies, 26.04.2020 00:51