
Chemistry, 15.12.2019 17:31 Bianca1203
Avolume of 25.0 ml of nitric acid, hno3, is titrated with .12 m naoh. to completely neutralize the acid 10.0 ml of naoh must be added. what is the molarity of nitric acid is

Answers: 1


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 08:30, omoaye
Identify one disadvantage to each of the following models of electron configuration: -dot structures -arrow and line diagrams -written electron configurations type in your answer below. (answer) -dot structures do not show the distribution of electrons in orbitals and take up a lot of space. -arrow and line diagrams take up a lot of space and make it difficult to count electrons. -written configurations make it easy to lose count of electrons and do not show the distribution of electrons in orbitals.
Answers: 3

Chemistry, 22.06.2019 16:50, Pookiev
Which of the following is an indication that a substance has undergone a chemical change? a. no new product has been formed. b. the color of the substance has not changed. c. the original constitute has not changed. d. the molecular structure has changed.
Answers: 1

Chemistry, 23.06.2019 07:30, shealynh52
Achemist at a pharmaceutical company is measuring equilibrium constants for reactions in which drug candidate molecules bind to a protein involved in cancer. the drug molecules bind the protein in a 1: 1 ratio to form a drug-protein complex. the protein concentration in aqueous solution at 25 ˚c is 1.74 x10-6 m . drug a is introduced into the protein solution at an initial concentration of 2.00 x10-6m. drug b is introduced into a separate, identical protein solution at an initial concentration of 2.00 x10-6m. at equilibrium, the drug a-protein solution has an a-protein complex concentration of 1.00 x10-6m, and the drug b solution has a b-protein complex concentration of 1.40 x10-6m. a. calculate the kc value for the a-protein binding reaction. b. calculate the kc value for the b-protein binding reaction. c. assuming that the drug that binds more strongly will be more effective, which drug is the better choice for further research?
Answers: 1
You know the right answer?
Avolume of 25.0 ml of nitric acid, hno3, is titrated with .12 m naoh. to completely neutralize the a...
Questions in other subjects:

History, 12.12.2020 21:30

Mathematics, 12.12.2020 21:30

Social Studies, 12.12.2020 21:30




Arts, 12.12.2020 21:30

Spanish, 12.12.2020 21:30


Mathematics, 12.12.2020 21:30

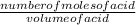
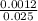 = 0.048M
= 0.048M

