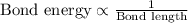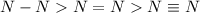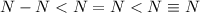Chemistry, 21.11.2019 22:31 kimhayleeshook50
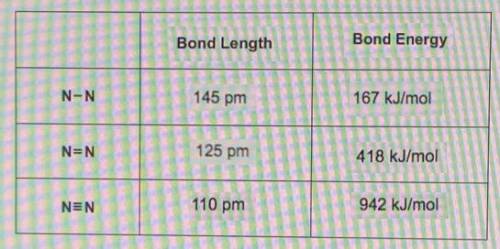
Complete the table of bond energies and bond lengths.
need answer !

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 17:00, sillslola816oxb5h7
An aqueous solution of hydroiodic acid is standardized by titration with a 0.186 m solution of calcium hydroxide. if 26.5 ml of base are required to neutralize 20.3 ml of the acid, what is the molarity of the hydroiodic acid solution? m hydroiodic acid
Answers: 1

Chemistry, 21.06.2019 18:00, paolaviviana
What does earth’s rotation on its axis cause? the tides night and day passing of years phases of the moon
Answers: 1

Chemistry, 22.06.2019 16:00, anaalashay
How will the volume of a gas be affected if the pressure is tripled, but the temperature remains the same?
Answers: 3

Chemistry, 22.06.2019 20:20, carcon2019
Nitric acid can be formed in two steps from the atmospheric gases nitrogen and oxygen, plus hydrogen prepared by reforming natural gas. in the first step, nitrogen and hydrogen react to form ammonia: (g) (g) (g) in the second step, ammonia and oxygen react to form nitric acid and water: (g) (g) (g) (g) calculate the net change in enthalpy for the formation of one mole of nitric acid from nitrogen, hydrogen and oxygen from these reactions. round your answer to the nearest .
Answers: 3
You know the right answer?
Complete the table of bond energies and bond lengths.
need answer !
...
need answer !
...
Questions in other subjects:


Mathematics, 07.01.2021 21:00



Advanced Placement (AP), 07.01.2021 21:00

Mathematics, 07.01.2021 21:00

Mathematics, 07.01.2021 21:00


Mathematics, 07.01.2021 21:00

Mathematics, 07.01.2021 21:00