Chemistry, 08.12.2019 08:31 janiyaf8941
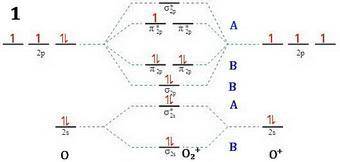
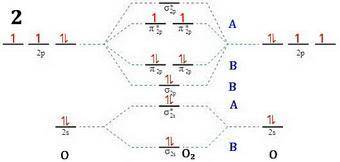
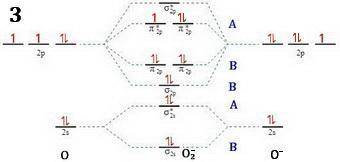
Apply molecular orbital theory to predict which species has the strongest bond. apply molecular orbital theory to predict which species has the strongest bond. o+2 o−2 o2 all bonds are equivalent according to molecular orbital theory.

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 17:40, Calumworthy6046
What are the resulting coefficients when you balance the chemical equation for the combustion of ethane, c2h6? in this reaction, ethane is burned in the presence of oxygen (o2) to form carbon dioxide (co2) and water (h2o). (g)+(g)→(g)+(g)
Answers: 1

Chemistry, 22.06.2019 08:20, pilarmonsivais
What is the formula for the compound dinitrogen pentoxide? a. n4o5 b. n5o4 c. n4o6 d. n5o2 e. n2o5
Answers: 3


Chemistry, 22.06.2019 22:30, pookie879
You just calculated that the heat of fusion for chloromethane is 6400 j/mol. the heat of fusion for hydrogen is 120 j/mol.? which of the following account for this difference? more than one correcta. chloromethane can absorb more energy at the same temperature. b. hydrogen has stronger intermolecular forces than chloromethane. c. hydrogen molecules can pack more closely than chloromethane molecules. d. chloromethane experiences dipole-dipole interactions. e. chloromethane has a higher molar mass than hydrogen.
Answers: 3
You know the right answer?
Apply molecular orbital theory to predict which species has the strongest bond. apply molecular orbi...
Questions in other subjects:

Biology, 19.10.2019 17:30

English, 19.10.2019 17:30





Mathematics, 19.10.2019 17:30



Social Studies, 19.10.2019 17:30