Hello!
The answer is:
The empirical formula is the option B. 
Why?
The empirical formula of a compound is the simplest formula that can be written. On the opposite, the molecular formula involves a variant of the same compound, but it can be also simplified to an empirical formula.

We are looking for a formula that cannot be simplified by dividing the number of molecules/atoms that conforms the compound.
Let's discard option by option in order to find which formula is an empirical formula (cannot be simplified)
A. 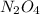
It's not an empirical formula, it's a molecular formula since it can be obtained by multiplying the empirical formula of the same compound.
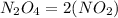
B. 
It's an empirical formula since it cannot be obtained by the multiplication of a whole number and the simplest formula. It's the simplest formula that we can find of the compound.
C. 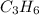
It's not an empirical formula, it's a molecular formula since it can be obtained by multiplying the empirical formula of the same compound.
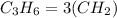
D. 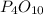
It's not an empirical formula, it's a molecular formula since it can be obtained by multiplying the empirical formula of the same compound.
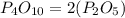
Hence, the empirical formula is the option B. 
Have a nice day!















