
Chemistry, 28.01.2020 13:40 heyyyyy3922
As with any combustion reaction, the products of combusting a hydrocarbon fuel (cxhy) with oxygen (o2) are carbon dioxide (co2) and water (h2o).
a mass of 15.51 g for an unknown fuel was combusted in a reaction vessel containing an unknown amount of oxygen. at the end of the reaction, there still remained 15.46 g of the fuel as well as 0.0817 g of water and 0.1497 g of carbon dioxide. the oxygen was completely consumed during the reaction.
how many molecules of oxygen gas were initially present in the reaction vessel?

Answers: 1


Other questions on the subject: Chemistry



Chemistry, 22.06.2019 03:00, bchagnard2122
Compare the valence electron configuration of the nobles gas elements seen here. what statement is correct?
Answers: 2
You know the right answer?
As with any combustion reaction, the products of combusting a hydrocarbon fuel (cxhy) with oxygen (o...
Questions in other subjects:


Mathematics, 20.07.2019 07:00




Biology, 20.07.2019 07:00




History, 20.07.2019 07:00

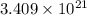
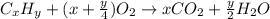
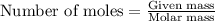
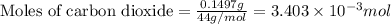
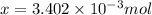
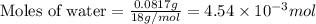
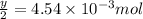
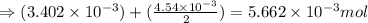
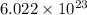 number of molecules.
number of molecules.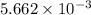 moles of oxygen will contain =
moles of oxygen will contain = 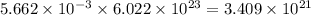 molecules.
molecules.

