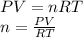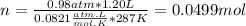Chemistry, 04.02.2020 01:53 sebastiantroysmith
Quickly !
part 1. determine the molar mass of a 0.458-gram sample of gas having a volume of 1.20 l at 287 k and 0.980 atm. show your work. part 2. if this sample was placed under extreme pressure, describe how the actual volume would compare to the predicted volume. explain your answer.

Answers: 2


Other questions on the subject: Chemistry


Chemistry, 23.06.2019 17:00, bradenjesmt1028
An unknown substance was put in a container. the substance filled up the container from bottom up, taking half of the space in the container. what is most likely the state of matter of the substance? gas liquid plasma solid
Answers: 3

Chemistry, 23.06.2019 18:20, ccelestineiyanoup3y8
Consider the following system at equilibrium. caco3(s) = ca2+(aq) + co32-(aq) the addition of which compound will cause a shift in equilibrium because of a common ion effect? occia co2 cuso4 na2co3
Answers: 3

Chemistry, 23.06.2019 19:30, gabriel5575
Which of the following is true about science? select one: a. it is not influenced by social conditions. b. it is not determined by external local factors. c. political conditions are unable to influence it. d. economic concerns may prevent it from solving problems.
Answers: 1
You know the right answer?
Quickly !
part 1. determine the molar mass of a 0.458-gram sample of gas having a volume of 1...
part 1. determine the molar mass of a 0.458-gram sample of gas having a volume of 1...
Questions in other subjects:


History, 12.09.2019 06:20


Mathematics, 12.09.2019 06:20

Mathematics, 12.09.2019 06:20

Geography, 12.09.2019 06:20


Mathematics, 12.09.2019 06:20

Mathematics, 12.09.2019 06:20

Mathematics, 12.09.2019 06:20