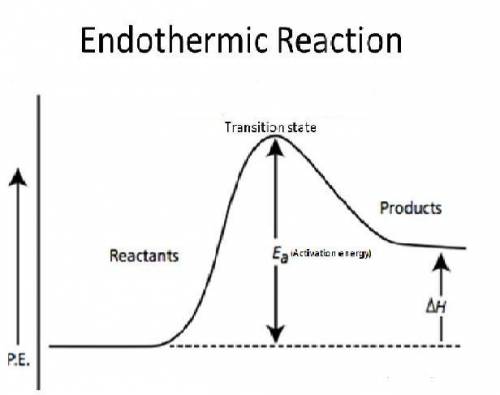
Why does every chemical reaction require a certain amount of activation energy?
a.
en...

Chemistry, 28.01.2020 07:31 kingdevin16
Why does every chemical reaction require a certain amount of activation energy?
a.
energy is released when the reactants begin to react.
b.
energy lost to the environment during the reaction must be replaced.
c.
forming the activated complex requires energy.
d.
the products have more potential energy than the activated complex.
e.
the reactants have less potential energy than the products.

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 08:30, itzhari101
In a chemical reaction at equilibrium, the rate of the forward reaction the rate of the reverse reaction. if the rate of the forward reaction more products are formed.
Answers: 1

Chemistry, 22.06.2019 14:00, jivsf
The two naturally occurring isotopes of chlorine are 35cl (34.969 amu, 75.77%) and 37cl (36.966 amu, 24.23%). the two naturally occurring isotopes of bromine are 79br (78.918 rm amu, 50.69%) and 81br (80.916 amu, 49.31%). chlorine and bromine combine to form bromine monochloride, brcl. 1. how many peaks will be present in a mass spectrum for brcl? the four combinations of molecule possible given these four isotopes are: 81br37cl, 81br35cl, 79br37cl, and 79br35cl. 2. what are the masses of the four different brcl molecules? express the masses using six significant figures, in decreasing numeric order (highest to lowest), separated by commas.
Answers: 3

Chemistry, 22.06.2019 21:00, lalaween098
What type of radiation is lead emitting in the following equation? alpha particles beta particles gamma rays
Answers: 3

Chemistry, 23.06.2019 04:31, saladdressing1425
Chemical engineering who specializes in negotiating for large purchases and instructing customers in use of the products are
Answers: 1
You know the right answer?
Questions in other subjects:

Biology, 29.09.2021 22:20

Mathematics, 29.09.2021 22:20







Chemistry, 29.09.2021 22:20