
Chemistry, 28.11.2019 07:31 Homepage10
When fully inflated a hot air balloon has a volume of 1.6 *10^5 l ( liter) an average temperature of 373k and 0 967 atm . 1) assuming that the air has an average molar mass of 29g/ mol what is the density of the air in the hot air balloon? p = g/l . (2) how does this compare with the density of air at stp ? (a) . it is less dense than at stp . (b) it is more dense than at stp .

Answers: 1


Other questions on the subject: Chemistry



Chemistry, 22.06.2019 13:00, rome58
Lab reagent, hypothesis test. a reference solution used as a lab reagent is purported to have a concentration of 5 mg/dl. six samples are taken from this solution and the following concentrations are recorded: (5.32, 4.88, 5.10, 4.73, 5.15, 4.75) mg/dl. these six measurements are assumed to be an srs of all possible measurements from solution. they are also assumed to have a standard deviation of 0.2, a normal distributin, and a mean concentration equal to the true concentration of the solution. carry out a significance test to determine whether these six measurements provide reliable evidence that the true concentration of the solution is actually not 5 mg/dl.
Answers: 1

Chemistry, 22.06.2019 15:00, tcapele252
‘which reaction would most likely require the use of an inert electrode?
Answers: 1
You know the right answer?
When fully inflated a hot air balloon has a volume of 1.6 *10^5 l ( liter) an average temperature of...
Questions in other subjects:

Mathematics, 03.06.2021 15:00

Mathematics, 03.06.2021 15:00


Chemistry, 03.06.2021 15:00


Geography, 03.06.2021 15:00



Mathematics, 03.06.2021 15:00

Mathematics, 03.06.2021 15:00


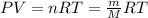
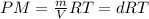
 )
)



