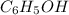Answers: 1


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 11:50, hadwell34
Which of the following statements about hybrid orbitals is or are true? choose all that apply. choose all that apply. under sp2 hybridization, the large lobes point to the vertices of an equilateral triangle. after an atom undergoes sp hybridization there is one unhybridized p orbital on the atom. the angle between the large lobes of sp3 hybrids is 109.5∘
Answers: 2


Chemistry, 22.06.2019 14:50, wcraig1998
Complete the following statements to describe solids, liquids, and gases. select the correct answer from each drop-down menu. a solid a definite volume and a definite shape. a liquid a definite volume and a definite shape. a gas a definite volume and a definite shape
Answers: 1
You know the right answer?
Achemist needs 22.0 grams of phenol, c6h5oh, for an experiment. how many molecules of phenol would t...
Questions in other subjects:



Mathematics, 19.05.2021 16:20

Mathematics, 19.05.2021 16:20


Mathematics, 19.05.2021 16:20

Mathematics, 19.05.2021 16:20



Mathematics, 19.05.2021 16:20

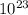 molecules in 22 grams of phenols.
molecules in 22 grams of phenols.
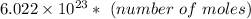
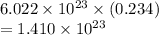
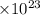 molecules.
molecules.