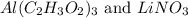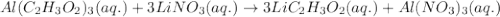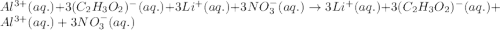Chemistry, 25.01.2020 16:31 brooklyn4932
Give the net ionic equation for the reaction (if any) that occurs when aqueous solutions of al(c2h3o2)3 and lino3 are mixed. give the net ionic equation for the reaction (if any) that occurs when aqueous solutions of al(c2h3o2)3 and lino3 are mixed. al3+(aq) + 3 no3-(aq) → al(no3)3(s) 3 li+(aq) + (c2h3o2)33-(aq) → li3(c2h3o2)3(s) al3+(aq) + 3 no3-(aq) + li+(aq) + c2h3o2-(aq) → al(no3)3(aq) + lic2h3o2(s) li+(aq) + c2h3o2-(aq) → lic2h3o2(s) no reaction occurs.

Answers: 2


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 22:00, Porciabeauty6788
The diagrams to the right show the distribution and arrangement of gas particles in two different containers. according to kinetic-molecular theory, which of the following statements is true? check all that apply. if the temperatures of both containers are equal, container a has greater pressure than container b. if the volume of container a decreased, its pressure would decrease. if the pressure in both containers is equal, container a has a lower temperature than container b.
Answers: 2

Chemistry, 22.06.2019 23:00, SophieCasey
What is the oxidation state of each individual carbon atom in c2o42−?
Answers: 1
You know the right answer?
Give the net ionic equation for the reaction (if any) that occurs when aqueous solutions of al(c2h3o...
Questions in other subjects:



Mathematics, 29.10.2019 04:31

Mathematics, 29.10.2019 04:31

Mathematics, 29.10.2019 04:31





History, 29.10.2019 04:31