Read the given equation.
2na + 2h2o → 2naoh + h2
during a laboratory experim...

Chemistry, 30.09.2019 08:30 winterblanco
Read the given equation.
2na + 2h2o → 2naoh + h2
during a laboratory experiment, a certain quantity of sodium metal reacted with water to produce sodium hydroxide and hydrogen gas. what was the initial quantity of sodium metal used if 6.30 liters of h2 gas were produced at stp?
a. 10.3 grams
b. 12.9 grams
c. 14.7 grams
d.15.2 grams

Answers: 2


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 09:40, kolibeilfuss
Sulfur dioxide and oxygen react to form sulfur trioxide during one of the key steps in sulfuric acid synthesis. an industrial chemist studying this reaction fills a 25.0l tank with 4.5 mol of sulfur dioxide gas and 4.5 mol of oxygen gas at 30.°c. he then raises the temperature, and when the mixture has come to equilibrium measures the amount of sulfur trioxide gas to be 1.4 mol. calculate the concentration equilibrium constant for the reaction of sulfur dioxide and oxygen at the final temperature of the mixture. round your answer to 2 significant digits.
Answers: 3

Chemistry, 22.06.2019 23:30, bxymichelle
With the largest atoms and the smallest number of valence electrons and with the smallest atoms and the greatest number of valence electrons are the most reactive. a. nonmetals; metals b. nonmetals; transition elements c. transition elements; metals d. metals; nonmetals
Answers: 3
You know the right answer?
Questions in other subjects:

Physics, 30.03.2020 19:19

Mathematics, 30.03.2020 19:19

Mathematics, 30.03.2020 19:19


History, 30.03.2020 19:19


Mathematics, 30.03.2020 19:19

History, 30.03.2020 19:19

Social Studies, 30.03.2020 19:19

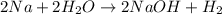
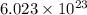 of particles and weighs equal to the molecular mass.
of particles and weighs equal to the molecular mass. is produced by 46 gram of sodium metal
is produced by 46 gram of sodium metal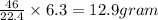 of sodium metal
of sodium metal

