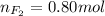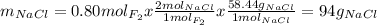Part 1. a chemist reacted 18.0 liters of f2 gas with nacl in the laboratory to form cl2 gas and naf. use the ideal gas law equation to determine the mass of nacl that reacted with f2 at 290. k and 1.5 atm.
f2 + 2nacl → cl2 + 2naf
part 2. explain how you would determine the mass of sodium chloride that can react with the same volume of fluorine gas at stp.

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 21:00, yousifgorgees101
The earth's moon is unusually large. two popular theories of the moon's origin include the "sister world" hypothesis, which states that the moon formed from the same materials as the earth, near enough to the earth that they fell into orbit around each other. a second theory is the "capture" hypothesis, in which the moon formed elsewhere in the solar system, and the earth's gravity pulled it into its orbit. studies of what the moon is made of indicate that some of its materials had to come from the earth or from the same area of the solar system where the earth had formed. at the same time, the moon does not contain much of the material that makes up the earth's core, so the moon could not have formed from the same materials as the earth. how do the two facts above affect the described theories of the moon's origin? a. they show that scientists will never agree on where the moon came from. b. they show that more experiments on moon formation need to be done. c. they show that no theory accounts for the existence of the moon. d. they show that neither theory is complete and entirely correct.
Answers: 1

Chemistry, 22.06.2019 03:50, sgslayerkingminecraf
Which of the following statements about acidic water is true? a. acid has no effect on the h, o molecules. b. the solution contains a larger number of oh ions than h, o ions. c. the solution contains a larger number of h, o ions than qh ions. d. the solution contains an equal number of h, o ions and oh ions. none of the above e.
Answers: 1


You know the right answer?
Part 1. a chemist reacted 18.0 liters of f2 gas with nacl in the laboratory to form cl2 gas and naf....
Questions in other subjects:




Mathematics, 04.12.2020 23:10

Computers and Technology, 04.12.2020 23:10





 →
→ 
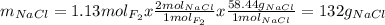
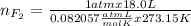 →
→