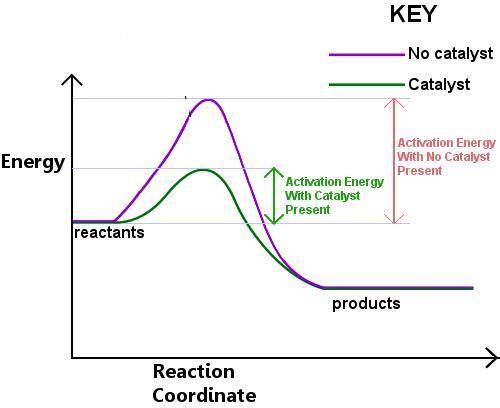
How does activation energy affect a chemical reaction ?
a. it increases the energy of...

Chemistry, 24.01.2020 17:31 kylahnicolewebb
How does activation energy affect a chemical reaction ?
a. it increases the energy of the reactants
b. it determines how difficult it is to start the reaction
c. it determines how much product can form
d. it decreases the energy of the products

Answers: 3


Other questions on the subject: Chemistry




Chemistry, 23.06.2019 00:30, Keemdadream13
If there are 3.5 moles of koh, how many moles of naoh can be produced? question 1 options: a)3.0 moles naoh b)3.5 moles naoh c)1 moles naoh d)9 moles naoh
Answers: 1
You know the right answer?
Questions in other subjects:



Health, 25.10.2019 03:43

Arts, 25.10.2019 03:43

Social Studies, 25.10.2019 03:43

Mathematics, 25.10.2019 03:43