Chemistry, 19.12.2019 11:31 erinloth123
Aph scale with ph values of some aqueous solutions. look at the figure showing the ph values of some familiar aqueous solutions. what is the difference between h+ concentration in an acidic solution such as lemon juice (ph 2) and a basic solution such as household bleach (ph 13)? a ph scale with ph values of some aqueous solutions. look at the figure showing the ph values of some familiar aqueous solutions. what is the difference between h+ concentration in an acidic solution such as lemon juice (ph 2) and a basic solution such as household bleach (ph 13)? the h+ concentration of lemon juice is higher than the h+ concentration of household bleach by a factor of 11. the h+ concentration of lemon juice is higher than the h+ concentration of household bleach by a factor of 1011 (100 billion). the h+ concentration of household bleach is higher than the h+ concentration of lemon juice by a factor of 1011 (100 billion).

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 18:10, hannacarroll2539
The enthalpy of formation of water is -285.8 kj/mol. what can be inferred from this statement?
Answers: 1


Chemistry, 22.06.2019 14:40, neonbluefaith
Which statement best describes the function of enzymes?
Answers: 1
You know the right answer?
Aph scale with ph values of some aqueous solutions. look at the figure showing the ph values of some...
Questions in other subjects:






History, 01.10.2019 19:30



Mathematics, 01.10.2019 19:30

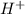 concentration of lemon juice is higher than the
concentration of lemon juice is higher than the 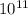 (100 billion)
(100 billion)![pH=-log[H^+]](/tpl/images/0425/9876/15713.png)
![2=-log[H^+]](/tpl/images/0425/9876/a01ca.png)
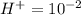
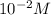 of
of ![13=-log[H^+]](/tpl/images/0425/9876/f4c07.png)
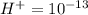
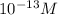 of
of