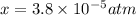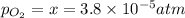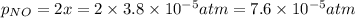Chemistry, 22.06.2019 20:00 denaemarie02
Nitrogen dioxide decomposes according to the reaction 2 no2(g) ⇌ 2 no(g) + o2(g) where kp = 4.48 × 10−13 at a certain temperature. if 0.70 atm of no2 is added to a container and allowed to come to equilibrium, what are the equilibrium partial pressures of no(g) and o2(g)

Answers: 2


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 20:00, emilyswinge4421
Listenbase your answer to the question on the information below. nuclear radiation is harmful to living cells, particularly to fast-growing cells, such as cancer cells and blood cells. an external beam of the radiation emitted from a radioisotope can be directed on a small area of a person to destroy cancer cells within the body. cobalt-60 is an artificially produced radioisotope that emits gamma rays and beta particles. one hospital keeps a 100.0-gram sample of cobalt-60 in an appropriate, secure storage container for future cancer treatment. which choice represents the correct product for the beta decay of the co-60? fe-60ni-60fe-61ni-61
Answers: 2

Chemistry, 22.06.2019 23:00, lulprettyb
What is the most common reason for matter changing its state?
Answers: 1
You know the right answer?
Nitrogen dioxide decomposes according to the reaction 2 no2(g) ⇌ 2 no(g) + o2(g) where kp = 4.48 × 1...
Questions in other subjects:




Mathematics, 16.12.2021 06:10




Chemistry, 16.12.2021 06:10


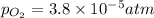
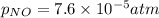
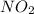 = p = 0.70 atm
= p = 0.70 atm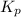 for the reaction
for the reaction 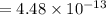
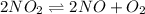
![K_p=\frac{[2x]^2[x]}{[p-2x]^2}](/tpl/images/0005/2998/5610b.png)
![4.48\times 10^{-13}=\frac{4x^3}{[0.70-2x]^2}](/tpl/images/0005/2998/9a087.png)