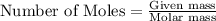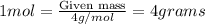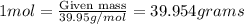For a: The number of atoms of both the gases remains the same.
For b: The number of grams of 1 mole of argon is more than 1 mole of helium by 35.95 grams.
Explanation:
For a:
To calculate the number of atoms, we use mole concept.
According to mole concept:
1 mole of an element contains 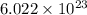 number of atoms.
number of atoms.
We are given two gases of same number of moles.
Therefore, 1 mole of helium gas will contain 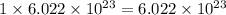 number of atoms.
number of atoms.
and, 1 mole of argon gas will contain 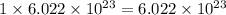 number of atoms.
number of atoms.
Hence, number of atoms of both the given gases remains the same.
For 2:
As, the molar mass of both the gases are different, so their masses in 1 mole will be different.
To calculate the number of moles, we use the equation:
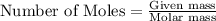
Calculating the mass of Helium in 1 mole:
Molar mass of helium = 4 g/mol
Putting the values in above equation, we get:
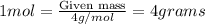
Calculating the mass of Argon in 1 mole:
Molar mass of Argon = 39.95 g/mol
Putting the values in above equation, we get:
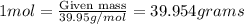
Calculating the difference of masses of both the gases = (39.95 - 4)g = 35.95g
Therefore, the mass of argon is more than mass of helium by 35.95 grams.

















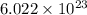 number of atoms.
number of atoms. 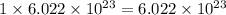 number of atoms.
number of atoms.