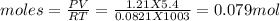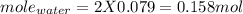Chemistry, 24.06.2019 19:30 luluhawkins
You form water vapor by mixing oxygen and hydrogen at 730°c in a 5.4-liter container. this is the equation for the reaction: o2(g) + 2h2(g) → 2h2o(g). the partial pressure of oxygen before the reaction is 122.3 kilopascals, and there is excess hydrogen. how many moles of water are formed? the reaction produces moles of water.

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 15:00, raeprince9213
Which of the following is the correct formula for copper (i) sulfate trihydrate? cuso4 · 3h2o cuso4(h2o)3 cu2so4(h2o)3 cu2so4 · 3h2o
Answers: 1

Chemistry, 23.06.2019 02:00, hermesrobles
Which would freeze at a higher temperature: the great salt lake or lake tahoe? a. lake tahoe would freeze at a higher temperature. b. the great salt lake would freeze at a higher temperature. c. both lakes would freeze at the same temperature.
Answers: 2

Chemistry, 23.06.2019 04:00, zakarycrane8101
If you are told to get 100 ml of stock solution to use to prepare smaller size sample for an experiment, which piece of glassware would you use?
Answers: 3
You know the right answer?
You form water vapor by mixing oxygen and hydrogen at 730°c in a 5.4-liter container. this is the eq...
Questions in other subjects:



Social Studies, 09.11.2019 03:31

Computers and Technology, 09.11.2019 03:31

Mathematics, 09.11.2019 03:31




Mathematics, 09.11.2019 03:31

Mathematics, 09.11.2019 03:31

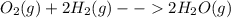
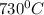 =1003K
=1003K