
Chemistry, 25.06.2019 05:00 chloeann5397
Which statement best identifies and describes the oxidizing agent in the reaction? 2al + 3br2 → 2albr3 al is the oxidizing agent because its oxidation number increases. al is the oxidizing agent because its oxidation number decreases. br2 is the oxidizing agent because its oxidation number increases. br2 is the oxidizing agent because its oxidation number decreases.

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 04:30, KarenH3512
Use the drop-down menus to answer each question. which runner finished the 100 m race in the least amount of time? which runner stopped running for a few seconds during the race? at what distance did anastasia overtake chloe in the race?
Answers: 1

Chemistry, 22.06.2019 12:00, daytonalive83481
Ican determine the molar mass of an element by looking on the under the atomic mass for the element. for example the molar mass of phosphorus is 30.974 grams/mole. avogadro’s number tells me the amount of representative particles in 1 mole of any substance. this means 12.011 gram sample of carbon and a 32.0 gram sample of sulfur have the same number of atoms.
Answers: 1

Chemistry, 22.06.2019 13:50, awesomegamergurl13
What happens when an atom of sulfur combines with two atoms of chlorine to produce sci2? a. each chlorine atom shares a pair of electrons with the sulfur atom. b. an electron is transferred from each chlorine atom to the sulfur atom. c. an electron is transferred from the sulfur atom to each chlorine atom. d. each chlorine atom shares all its valence electrons with the sulfur atom.
Answers: 2

You know the right answer?
Which statement best identifies and describes the oxidizing agent in the reaction? 2al + 3br2 → 2al...
Questions in other subjects:

Social Studies, 19.07.2020 02:01

Mathematics, 19.07.2020 02:01

Mathematics, 19.07.2020 02:01


Mathematics, 19.07.2020 02:01

Mathematics, 19.07.2020 02:01

Mathematics, 19.07.2020 02:01

Mathematics, 19.07.2020 02:01


English, 19.07.2020 02:01

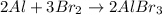
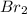 is the oxidizing agent because its oxidation number decreases.
is the oxidizing agent because its oxidation number decreases.

