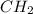Chemistry, 26.06.2019 08:30 theirishgold42
Which pair shares the same empirical formula? a. c2h2 and c6h6 b. c2h2 and c2h4 c. ch2 and c6ch6 d. ch and c2h4

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 08:30, omoaye
Identify one disadvantage to each of the following models of electron configuration: -dot structures -arrow and line diagrams -written electron configurations type in your answer below. (answer) -dot structures do not show the distribution of electrons in orbitals and take up a lot of space. -arrow and line diagrams take up a lot of space and make it difficult to count electrons. -written configurations make it easy to lose count of electrons and do not show the distribution of electrons in orbitals.
Answers: 3


Chemistry, 22.06.2019 17:00, jazmine8194
Reduction is a reaction which results in a in electrons and a in positive charge of the atom or ion 1) a- loss 1) b- gain 2) a-increase 2) b-decrease
Answers: 1

Chemistry, 22.06.2019 22:00, shaylasimonds587
The volume of an unknown substance in a sealed glass jar is 50 milliliters. the volume of the jar is 200 milliliters. which state of matter could the substance be?
Answers: 2
You know the right answer?
Which pair shares the same empirical formula? a. c2h2 and c6h6 b. c2h2 and c2h4 c. ch2 and c6ch6 d....
Questions in other subjects:





World Languages, 17.10.2019 02:00





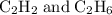 .
.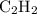 has the empirical formula =
has the empirical formula = 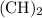 = CH
= CH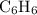 has the empirical formula =
has the empirical formula = 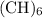 = CH
= CH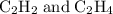 :
: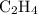 has the empirical formula =
has the empirical formula = 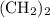 =
= 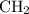
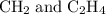 :
: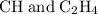 :
: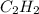 and
and 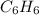
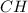 and
and 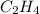 : Empirical formula for
: Empirical formula for