
Water is known as the universal solvent. one reason water makes a good solvent is because it is composed of polar molecules. what makes a water molecule a polar molecule? a. the water molecule never develops a charge and is always neutral. water molecules are rod-shaped. b. the water molecule has a negative charge at cold temperatures. c. water molecules have a positive charge on one end and a negative charge on the other.

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 17:00, kathleendthomas
Look at the reaction below: ca(hco3)2 --> caco3 + co2 + h2o first, balance the reaction. once balanced, use dimensional analysis or another method to find out how many moles of carbon dioxide will be produced if we start with 16.5 moles of calcium bicarbonate (calcium hydrogen carbonate). = mol of co2 number needs to be reported to three significant figures.
Answers: 1

Chemistry, 22.06.2019 23:30, bxymichelle
With the largest atoms and the smallest number of valence electrons and with the smallest atoms and the greatest number of valence electrons are the most reactive. a. nonmetals; metals b. nonmetals; transition elements c. transition elements; metals d. metals; nonmetals
Answers: 3

You know the right answer?
Water is known as the universal solvent. one reason water makes a good solvent is because it is comp...
Questions in other subjects:

Mathematics, 13.10.2019 01:00

Mathematics, 13.10.2019 01:00



History, 13.10.2019 01:00



English, 13.10.2019 01:00

Mathematics, 13.10.2019 01:00

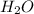 .
. 

