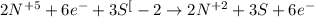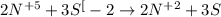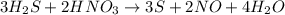Chemistry, 26.06.2019 21:30 trinitieu66
Consider this reaction: h2s + hno3 s + no + h2o reduction half-reaction: n+5 + 3e- n+2 oxidation half-reaction: s-2 s + 2e- after balancing the chemical equation, what are the coefficients for these compounds?

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 08:00, wizz4865
Why is the bond angle in a water molecule less than the bond angle of methane? a. the central oxygen atom in water has two lone pairs of electrons, whereas the central carbon atom in methane has no lone pairs. b. the central hydrogen atom in water has one lone pair of electrons, whereas the central carbon atom in methane has two lone pairs. c. the central oxygen atom in water has four lone pairs of electrons, whereas the central carbon atom in methane has only one lone pair. d. the central oxygen atom exerts more repulsive force on surrounding atoms than the central carbon atom in methane does. reset next
Answers: 2

Chemistry, 23.06.2019 03:50, KAITLYN007
Show how to convert the temperature 84.7° c to kelvin. include all steps and label the final answer.
Answers: 1

Chemistry, 23.06.2019 05:30, choatefarmsus
Awhite powder is added to a solution. the images show observations made before the powder is added, just after the powder has been added, and a little while later. (the liquid in the small beaker is phenol red solution.) what evidence shows that a chemical change has taken place?
Answers: 1

Chemistry, 23.06.2019 11:50, natorihill629
Charles's law describes the relationship of the volume and temperature of gas at a constant mass and pressure. according to this law, what would happen to the temperature of the gas if its volume decreased from 1.0 l to 0.50 l?
Answers: 3
You know the right answer?
Consider this reaction: h2s + hno3 s + no + h2o reduction half-reaction: n+5 + 3e- n+2 oxidation h...
Questions in other subjects:










Mathematics, 07.11.2019 21:31

 = 3
= 3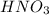 = 2
= 2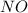 = 2
= 2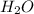 = 4
= 4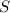 = 3
= 3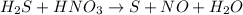
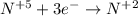 ...(1)
...(1)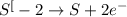 ...(2)
...(2)