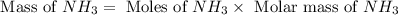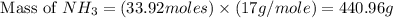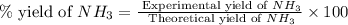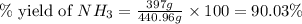Chemistry, 27.06.2019 20:30 elijahlylejamez45
25 i got the #1, just not #2 and #3. an industrial chemical company has opened a new plant that will produce ammonia (nh3). hydrogen and nitrogen gases are reacted to produce the ammonia. for the first batch of ammonia production, 475 g of nitrogen is reacted with excess hydrogen, and 397 g of ammonia are produced. • write the balanced equation for the formation of ammonia from hydrogen and nitrogen.  2nh3 • calculate the theoretical yield of ammonia. work must be shown to earn credit. • calculate the percent yield for the ammonia production. work must be shown to earn credit.
2nh3 • calculate the theoretical yield of ammonia. work must be shown to earn credit. • calculate the percent yield for the ammonia production. work must be shown to earn credit.

Answers: 1


Other questions on the subject: Chemistry



You know the right answer?
25 i got the #1, just not #2 and #3. an industrial chemical company has opened a new plant that wil...
Questions in other subjects:

French, 22.10.2020 21:01


Mathematics, 22.10.2020 21:01



English, 22.10.2020 21:01


Mathematics, 22.10.2020 21:01

History, 22.10.2020 21:01

Mathematics, 22.10.2020 21:01

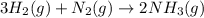
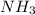 gas = 440.96 g
gas = 440.96 g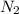 = 475 g
= 475 g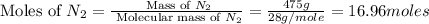
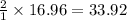 moles of
moles of