
Chemistry, 28.06.2019 02:30 jobucks9976
Fe3+(aq) (yellow) + scn-(aq) (colorless) fescn2+(aq) (blood-red) chloride ions are colorless. potassium ions are also colorless. the above equilibrium can be created by mixing an iron (iii) chloride solution with a potassium thiocyanate solution. based on this information and the colors in the equilibrium above answer the first two questions. 1. what color would an fecl3 solution be? 2. what color would a kscn solution be? 3. what color do you get when you mix fecl3 and kscn as shown in the video for the control test tube?

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 07:40, caleb19moody
21. consider the following chemical reaction: n2+ o2 2 no if 10.0 g of n2 reacts with excess oxygen then how many grams of no can be formed? a) 10.7 g b) 21.4 g c) 32.9 g d) 42.8 g page 4 of 8
Answers: 2


Chemistry, 23.06.2019 15:00, Fangflora3
What do we call the rows on the periodic table? a. periodb. familyc. groupd. metals
Answers: 1
You know the right answer?
Fe3+(aq) (yellow) + scn-(aq) (colorless) fescn2+(aq) (blood-red) chloride ions are colorless. potass...
Questions in other subjects:




Mathematics, 21.05.2020 07:58



English, 21.05.2020 07:58



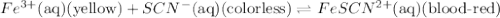
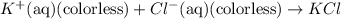
![FeCl_3+K(SCN)\rightleftharpoons KCl+[Fe(SCN)]Cl_2(blood-red)](/tpl/images/0025/5430/e3b23.png)
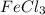 we will get blood red color solution of
we will get blood red color solution of ![[FeSCN]Cl_2](/tpl/images/0025/5430/ecffe.png) .
.

