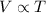Chemistry, 28.06.2019 09:30 rodriguezangelinaado
Asample of nitrogen gas occupies 2.49 l at 19.5°c. the volume of the container changes at constant pressure to 4.29 l. what temperature (in°c) must the gas be to attain this volume?

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 14:30, krystenlitten
Find the protons, electrons and neutrons for strontium with a mass of 83
Answers: 1


Chemistry, 22.06.2019 17:10, sophiaa23
)benzene and toluene form nearly ideal solutions. consider an equimolar solution of benzene and toluene. at 20 °c the vapour pressures of pure benzene and toluene are 9.9 kpa and 2.9 kpa, respectively. the solution is boiled by reducing the external pressure below the vapour pressure. calculate (i) the pressure when boiling begins, (ii) the composition of each component in the vapour, and (iii) the vapour pressure when only a few drops of liquid remain. assume that the rate of vaporization is low enough for the temperature to remain constant at 20 °c.
Answers: 1

Chemistry, 22.06.2019 19:00, hmontalvo22
How many moles are contained in 5.6 l of h2 at stp
Answers: 3
You know the right answer?
Asample of nitrogen gas occupies 2.49 l at 19.5°c. the volume of the container changes at constant p...
Questions in other subjects:

History, 02.03.2021 22:00

Computers and Technology, 02.03.2021 22:00

Mathematics, 02.03.2021 22:00

English, 02.03.2021 22:00

Mathematics, 02.03.2021 22:00

Mathematics, 02.03.2021 22:00

Mathematics, 02.03.2021 22:00


Mathematics, 02.03.2021 22:00