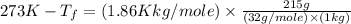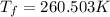Chemistry, 29.06.2019 23:30 FunnySkittle
Asolution is prepared by dissolving 215 grams of methanol, ch3oh, in 1000. grams of water. what is the freezing point of this solution? [the freezing point depression constant for water is 1.86°c/mole solute in 1000g of water]

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 14:50, ladybugperez05
Which of the following is most likely true about water in chemical systems? a) water dissolves nonpolar ionic compounds. b) water dissociates ionic compounds. c) water dissociates covalent molecules. d) water dissolves nonpolar covalent substances.
Answers: 1

Chemistry, 22.06.2019 20:30, lexibyrd120
Water undergoes a large change in density at 0 ∘ c as it freezes to form ice. calculate the percent change in density that occurs when liquid water freezes to ice at 0 ∘ c given that
Answers: 2

Chemistry, 23.06.2019 01:50, UncleVictor5188
Ablock of aluminum is dropped into a graduated cylinder with an initial volume of water at 75ml and the volumes rises to 90ml. if the block has a mass of 40.5 g what is its density ?
Answers: 1
You know the right answer?
Asolution is prepared by dissolving 215 grams of methanol, ch3oh, in 1000. grams of water. what is t...
Questions in other subjects:

Mathematics, 03.07.2019 11:00

Mathematics, 03.07.2019 11:00








Mathematics, 03.07.2019 11:00

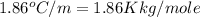
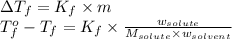
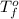 = freezing point of water =
= freezing point of water = 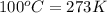
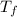 = freezing point of solution
= freezing point of solution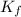 = freezing point constant
= freezing point constant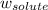 = mass of solute
= mass of solute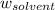 = mass of solvent
= mass of solvent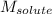 = molar mass of solute
= molar mass of solute