
Chemistry, 30.06.2019 00:00 Kstearns694
The equilibrium constant for the formation of ammonia from nitrogen and hydrogen is 1.6 × 102. what is the form of the equilibrium constant?

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 16:30, leilanimontes714
Asample of silver (with work function ? = 4.52 ev) is exposed to an ultraviolet light source (? = 200 nm), which results in the ejection of photoelectrons. what changes will be observed if: silver is replaced with copper (? = 5.10 ev) more photoelectrons ejected no photoelectrons are emitted fewer photoelectrons ejected more energetic photoelectrons (on average) less energetic photoelectrons (on average)
Answers: 3

Chemistry, 21.06.2019 22:50, Catracho3619
Blank allows you to do calculations for situations in which only the amount of gas is constant a)boyle's law b)combined gas law c)ideal gas law d)dalton's law
Answers: 1

Chemistry, 22.06.2019 08:30, itzhari101
In a chemical reaction at equilibrium, the rate of the forward reaction the rate of the reverse reaction. if the rate of the forward reaction more products are formed.
Answers: 1
You know the right answer?
The equilibrium constant for the formation of ammonia from nitrogen and hydrogen is 1.6 × 102. what...
Questions in other subjects:

History, 22.09.2021 03:00





Mathematics, 22.09.2021 03:00



Mathematics, 22.09.2021 03:00

Mathematics, 22.09.2021 03:00

![\frac{[NH_3]^2}{[H_2]^3[N_2]}](/tpl/images/0032/8317/ef503.png)
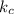
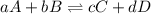
![k_c=\frac{[C]^c[D]^d}{[A]^a[B]^b}](/tpl/images/0032/8317/c796b.png)
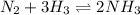
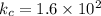
![k_c=\frac{[NH_3]^2}{[H_2]^3[N_2]}](/tpl/images/0032/8317/3f630.png)
![1.6\times 10^2=\frac{[NH_3]^2}{[H_2]^3[N_2]}](/tpl/images/0032/8317/d741a.png)


