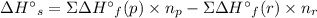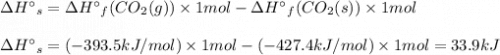Chemistry, 30.06.2019 07:00 ineedhelp2285
Use standard enthalpies of formation to calculate the change in enthalpy for dry ice sublimation. (the î´hâf for co2(s) is - 427.4kj/mol).

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 04:40, khan2491
Silver tarnishes as silver metal reacts with hydrogen sulfide, h2s, in the air. in this reaction, dark silver sulfide, au2s, covers the surface of silver. when silver is polished, this coating of silver sulfide can be removed from the surface. this makes the silver shiny again. enter the coefficients that balance the tarnishing reaction equation. (type 1 for no coefficient.)
Answers: 2



Chemistry, 22.06.2019 18:00, AdoNice
Many pharmaceutical drugs are organic compounds that were originally synthesized in the laboratory. which two scientific disciplines are bridged by pharmaceutical drugs? chemistry and forensics chemistry and medicine biology and forensics biology and criminology
Answers: 2
You know the right answer?
Use standard enthalpies of formation to calculate the change in enthalpy for dry ice sublimation. (t...
Questions in other subjects:


Spanish, 21.04.2021 02:10



French, 21.04.2021 02:10