
Chemistry, 30.06.2019 10:00 francisco42002
How would increasing the original concentration of a weak acid affect its ka and pka? a. ka and pka would increase. b. ka and pka would decrease. c. ka would increase and pka would decrease. d. ka would decrease and pka would increase. e. ka and pka would both stay the same.

Answers: 1


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 17:40, zayo8468
Experiment: effect of solution concentration on reaction rate you have learned that as the concentration of reactants increases, there will most likely be a greater number of collisions, and hence increase the rate of a reaction. in this experiment, you will see a demonstration of this, with a twist. there will be three reactions going on in this experiment. objectives determine how solution concentration can affect the rate of a reaction. the first reaction will be a reaction of the iodide ion (i-1) with hydrogen peroxide (h2o2) in an acidic solution. this reaction produces a slightly orange solution. in our experiment, we will add some orange food coloring to make this solution more orange. 2 h+ (aq) + 2 i- (aq) + h2o2 (aq) ⟶ i2 (aq) + 2 h2o (l) the next reaction will be between the iodine and starch i2 + starch ⟶ i2-starch complex (blue-black) so, when starch is added to the iodine solution made from the first reaction, the solution will turn black immediately, so it is difficult to find the rate of reaction. in order to be able to time this reaction, you will slow it down with another reaction. adding ascorbic acid will react with the iodine, reducing the concentration of the iodine available to react with the starch. c6h8o6 (aq) + i2 (aq) ⟶ 2i- (aq) + c6h6o6 (aq) + 2 h+ (aq) when the ascorbic acid is used up, the remaining iodine molecules can react with the starch and form the black color. the more ascorbic acid you add, the slower the reaction to form the iodine-starch complex will be. use your data and observations to complete the assignment. analysis and conclusions submit your data and the answers to these questions in the essay box below. what was your hypothesis? plot your data as drops of ascorbic acid vs. time. as the concentration of ascorbic acid was increased, did the rate of the formation of the iodine-starch complex increase or decrease? explain your answer in terms of the chemical reactions involved. was your hypothesis correct? make a general rule about the effects of concentration of reactants on reaction rates. for practice, the molecular formula for ascorbic acid is c6h8o6, and you used 6 g in this experiment, calculate the molarity of the ascorbic acid. now calculate the concentration in moles per drop (assume 1 ml = 20 drops).
Answers: 3

Chemistry, 22.06.2019 22:30, kiera2599
3.09 lab: reaction of metals 1 which combinations of substances resulted in a chemical change? for each metal that participated in a chemical change, write the type of metal it is, based on your examination of the periodic table. were there any metallic compounds that did not react with either the acid or the base? write the type of metal, based on your examination of the periodic table. make a general statement about the reactivity of the metals in this experiment.
Answers: 1

Chemistry, 23.06.2019 16:50, isyssjones42
Equal volumes of h2 and o2 are placed in a balloon and then ignited. assuming that the reaction goes to completion, which gas will remain in excess? 2h2 + o2 → 2h2o a. both are in equal proportion b. oxygen c. water d. hydrogen
Answers: 1
You know the right answer?
How would increasing the original concentration of a weak acid affect its ka and pka? a. ka and pk...
Questions in other subjects:

Mathematics, 24.07.2021 01:00

History, 24.07.2021 01:00


Arts, 24.07.2021 01:00

Mathematics, 24.07.2021 01:00


Chemistry, 24.07.2021 01:00

Mathematics, 24.07.2021 01:00

Mathematics, 24.07.2021 01:00

Computers and Technology, 24.07.2021 01:00

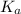 would increase and
would increase and 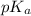 would decrease.
would decrease.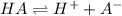
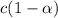
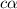
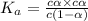
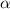 is very very small , the expression will be,
is very very small , the expression will be,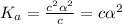
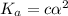 As
As 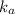 is directly proportional to concentration, the value of
is directly proportional to concentration, the value of 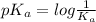
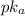 would decrease.
would decrease.

