Chemistry, 30.06.2019 12:00 clinton1790
What is the enthalpy for reaction 1 reversed? reaction 1 reversed: 2co2 + 3h2o→c2h5oh + 3o2 express your answer numerically in kilojoules per mole?

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 08:40, alexisbcatlett14
Which statement can best be concluded from the ideal gas law?
Answers: 2

Chemistry, 22.06.2019 14:30, Dreynolds1667
100 grams of molten lead (600°c) is used to make musket balls. if the lead shot is allowed to cool to room temperature (21°c), what is the change in entropy (in j/k) of the lead? (for the specific heat of molten and solid lead use 1.29 j/g⋅°c; the latent heat of fusion and the melting point of lead are 2.45 × 104 j/kg and 327°c, respectively.)
Answers: 1


Chemistry, 22.06.2019 20:00, denaemarie02
Nitrogen dioxide decomposes according to the reaction 2 no2(g) ⇌ 2 no(g) + o2(g) where kp = 4.48 × 10−13 at a certain temperature. if 0.70 atm of no2 is added to a container and allowed to come to equilibrium, what are the equilibrium partial pressures of no(g) and o2(g)
Answers: 2
You know the right answer?
What is the enthalpy for reaction 1 reversed? reaction 1 reversed: 2co2 + 3h2o→c2h5oh + 3o2 expres...
Questions in other subjects:


Mathematics, 24.10.2020 06:20

Biology, 24.10.2020 06:20

Biology, 24.10.2020 06:20

Mathematics, 24.10.2020 06:20

Mathematics, 24.10.2020 06:20

Biology, 24.10.2020 06:20


Geography, 24.10.2020 06:20

Mathematics, 24.10.2020 06:20

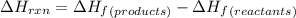
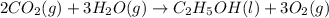
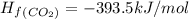
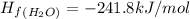
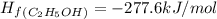
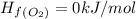
![\Delta H_{rxn}=\Delta H_f_{(C_2H_5OH)}+3\Delta H_f_{(O_2)}-[2\Delta H_f_{(CO_2)}+3\Delta H_f_{(H_2O)}]](/tpl/images/0034/7473/402a5.png)
![\Delta H_{rxn}=[-277.6+3(0)]-[2(-393.5)+3(-241.8)]kJ/mol](/tpl/images/0034/7473/4b8a1.png)