Chemistry, 30.06.2019 12:00 JuniperGalaxy
Sucrose (c12h22o11, table sugar) is oxidized in the body by o2 via a complex set of reactions that ultimately produces co2(g) and h2o(l) and releases 5.16 × 103 kj of heat per mole of sucrose. (a) write a balanced thermochemical equation for this reaction. include the physical state of each reactant and product. → enter your answer for δhrxn in scientific notation. δhrxn

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 05:50, mayamabjishovrvq9
Why doesn't heat added to water make the tempature rise above 100c
Answers: 2



Chemistry, 23.06.2019 01:30, babygirl091502
In what way do investigations build scientific knowledge? the results of investigations lead to questions that cannot be tested. they reflect the opinions and social values of scientists, ensuring valid information. the results of investigations lead to new questions, which lead to new investigations. they are not influenced by the research of earlier scientists, so they are able to address gaps in understanding. i
Answers: 1
You know the right answer?
Sucrose (c12h22o11, table sugar) is oxidized in the body by o2 via a complex set of reactions that u...
Questions in other subjects:



English, 27.08.2019 13:50

Chemistry, 27.08.2019 13:50

Mathematics, 27.08.2019 13:50

Spanish, 27.08.2019 13:50

Physics, 27.08.2019 13:50


Mathematics, 27.08.2019 13:50

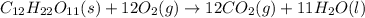
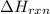 is represented in scientific notation as,
is represented in scientific notation as,