
Chemistry, 01.07.2019 01:30 iiwolfiexuni
The hydrogen atom is not actually electronegative enough to form bonds to xenon. were the xenon-hydrogen bond to exist, what would be the structure of xeh4? double-click any atom and type xe to change the label. draw the molecule by placing atoms on the grid and connecting them with bonds. show all lone pairs of electrons.

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 00:50, justabeachbum
If a reactant was removed, did the new equilibrium system shift to make more reactants or more products?
Answers: 1

Chemistry, 22.06.2019 10:00, nana54muller
Part 1: include important facts found through your research. part 2: include your visual display. include your summary of “the chemistry of water” from the national science foundation website. include your experiment. part 3: include responses to the reflection questions.
Answers: 1

Chemistry, 22.06.2019 18:00, tatemelliott
Which three statements represent the benefits of performing experiments using computer simulations?
Answers: 2

Chemistry, 22.06.2019 19:30, dorindaramirez0531
Which liquid (h2o, h2o + soap, or h2o + salt) has the strongest cohesion and adhesion? (need now plz)
Answers: 1
You know the right answer?
The hydrogen atom is not actually electronegative enough to form bonds to xenon. were the xenon-hydr...
Questions in other subjects:



Advanced Placement (AP), 16.11.2020 18:20

Mathematics, 16.11.2020 18:20

Biology, 16.11.2020 18:20

Mathematics, 16.11.2020 18:20

Mathematics, 16.11.2020 18:20


Mathematics, 16.11.2020 18:20

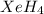 will be square-planar.
will be square-planar.


