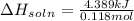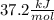Chemistry, 01.07.2019 03:00 paigeisawesome
Student conducts an experiment to determine the enthalpy of solution for lithium chloride dissolved in water. the student combines 5.00 grams of lithium chloride with 100.0 ml of distilled water. the initial temperature of the water is 23.0â°c and the highest temperature after mixing reaches 33.0â°c. assume a density of 1.00 g/ml and a specific heat of 4.18 .

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 01:40, georgehall3027
C3h8o3 - glycerol major species present when dissolved in water
Answers: 2

Chemistry, 22.06.2019 11:00, RidhaH
Which statement correctly identifies the scientific question and describes why the question is scientific? question 1 refers to the supernatural. question 2 reflects a moral or social value. question 3 refers to something that can be measured. question 4 reflects a question that can’t be observed.
Answers: 1

Chemistry, 22.06.2019 16:50, struckedblazing
Answer asap need it by wednesday morning calculate the ph of 0.02m hcl best answer will be brainliest
Answers: 1

Chemistry, 22.06.2019 17:20, phanuel642
The small bags of silica gel you often see in a new shoe box are placed there to control humidity. despite its name, silica gel is a solid. it is a chemically inert, highly porous, amorphous form of sio2. because water vapor readily adsorbs onto the surface of silica gel, it acts as a desiccant. despite not knowing mechanistic details of the adsorption of water onto silica gel, from the information provided you should be able to make an educated guess about the thermodynamic characteristics of the process. predict the signs for δg, δh, and δs for the adsorption of water.
Answers: 2
You know the right answer?
Student conducts an experiment to determine the enthalpy of solution for lithium chloride dissolved...
Questions in other subjects:



Engineering, 02.10.2020 22:01




French, 02.10.2020 22:01



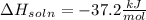

 = change in temperature
= change in temperature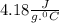
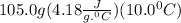
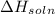 we convert q to kJ and divide by the moles of solute.
we convert q to kJ and divide by the moles of solute.
 = 4.389 kJ
= 4.389 kJ