Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 14:00, luisaareli6298
Calculate the energy required to ionize a hydrogen atom to an excited state where the electron is initially in the n = 5 energy level. report your answer in kilojoules
Answers: 1

Chemistry, 22.06.2019 21:30, imalexiscv
Plzz a sample of table sugar (sucrose, c12h22o11) has a mass of 7.801 g. ● a) calculate the number of moles of c12h22o11 in the sample b) calculate the number of moles of each element in c12h22o11 (number of moles of c, number of moles of h & number of moles of o) in the sample. (use your answer from part a as your starting point.) show your work and highlight your final answer. calculate the number of atoms of each element in c12h22o11 (number of atoms of c, number of atoms of h & number of atoms of o) in the sample. (use your answers from part b as your starting for each element.) show your work and highlight your final answer.
Answers: 1

Chemistry, 22.06.2019 23:00, autumperry3599
What is the chemical formula for dihydrogen monoxide
Answers: 2

Chemistry, 23.06.2019 07:30, klocke2001
Can you guys answer these questions i need it before 1: 00pm
Answers: 3
You know the right answer?
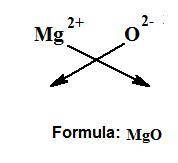
What’s an ionic compound for magnesium and oxide?...
Questions in other subjects:

Mathematics, 12.12.2020 17:00



Mathematics, 12.12.2020 17:00

Spanish, 12.12.2020 17:00


Mathematics, 12.12.2020 17:00


Mathematics, 12.12.2020 17:00

![[Ne]3s^{2}](/tpl/images/0043/8121/e0192.png)
![[He]2s^22p^4](/tpl/images/0043/8121/cdbb3.png)