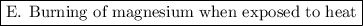 is a chemical property.
is a chemical property.
A characteristic feature of any substance that makes it different from the others is known as property.
Types of properties:
1. Physical properties
These are the properties that are measured without changing the chemical composition of the matter. These are used to describe and observe and matter. These can also be classified as physical and chemical properties, varying on their dependence. Some examples of physical properties are mass, volume, density, melting and boiling points.
2. Chemical properties:
These are the properties that come into account when any chemical reaction takes place. These cannot be observed by simple viewing or touching the substance. Some examples of chemical properties are toxicity, flammability, enthalpy of combustion, and chemical reactivity.
A. Density is defined as mass per unit volume. It can be measured without altering the chemical composition of matter so it is a physical property.
B. Melting point can be measured without altering the chemical composition of matter so it is a physical property.
C. Electrical Conductance is a physical property as it can be measured without changing the chemical composition of matter.
D. Color can be observed simply by just looking at the substance so it is a physical property.
E. When magnesium is exposed to heat, it burns and some new substances will be formed with the help of chemical reaction. The chemical composition of magnesium is changed so it is a chemical property.
Learn more:
Which of these is an extensive property?
Which of the phase changes is an exothermic change?
Answer details:
Grade: High School
Chapter: Physical and chemical properties
Subject: Chemistry
Keywords: chemical property, physical property, color, density, electrical conductance, heat, chemical composition, matter.



















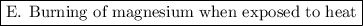 is a chemical property.
is a chemical property.


