
Chemistry, 03.07.2019 12:30 jalaholmes2027
Explain how fluorine and aluminum would exchange valence electrons to form an ionic compound.

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 02:00, bernicewhite156
Will give brainliest it is a lab from k12 here is the linkfor each metal that participated in a chemical change, write the type of metal it is, based on your examination of the periodic table. type your answer here. (score for question 3: of 5 points) were there any metallic compounds that did not react with either the acid or the base? write the type of metal, based on your examination of the periodic table. type your answer here. (score for question 4: of 5 points) make a general statement about the reactivity of the metals in this experiment. type your answer here.
Answers: 2

Chemistry, 22.06.2019 06:30, darrriannn7241
What is the correct lewis structure for chloroform chcl3
Answers: 1

Chemistry, 22.06.2019 08:30, aydenmasonc
Which statement describes james chadwick’s discovery.
Answers: 2

Chemistry, 22.06.2019 12:00, Alexislol7908
From the options provided for each element below, choose the properties that it may have based on its location in the periodic table fluorine (f): highly reactive nonmetal shiny a conductor
Answers: 1
You know the right answer?
Explain how fluorine and aluminum would exchange valence electrons to form an ionic compound....
Questions in other subjects:


Mathematics, 22.08.2020 23:01

History, 22.08.2020 23:01


Mathematics, 22.08.2020 23:01



Computers and Technology, 22.08.2020 23:01

Mathematics, 22.08.2020 23:01

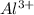 and three
and three  ions.
ions.



