Chemistry, 04.07.2019 22:00 jlayne0605
Which substance in this redox reaction is the oxidizing agent? cu + 2agno3 → 2ag + cu(no3)2 a. n b. agno3 c. cu d. no3− e. cu(no3)2

Answers: 2


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 02:40, gabrielolivas59
The difference between the atomic number of an element and the element’s atomic mass is the number of ions.
Answers: 3

Chemistry, 22.06.2019 05:30, MansellS5529
A3.37-mg sample of protein was chemically digested to convert its nitrogen into ammonia and then diluted to 100.0 ml. then 10.0 ml of this solution was placed in a 50-ml volumetric flask and treated with 5 ml of phenol solution plus 2 ml of sodium hypochlorite solution. the sample was diluted to 50.0 ml, and the absorbance at 625 nm was measured in a 1.00-cm cuvette and found to be 0.486. for reference, a standard solution was prepared from 10.0 mg of nh4cl (molar mass = 53.49 grams/mole) dissolved in 1.00 l of water. then 10.0 ml of this standard was placed in a 50-ml volumetric flask, treated in the same manner as the unknown, and the absorbance found to be 0.323. finally, a reagent blank was prepared using distilled water in place of unknown, it was treated in the same manner as the unknown, and the absorbance found to be 0.076. calculate the weight percent of nitrogen in the protein.
Answers: 1

Chemistry, 22.06.2019 07:00, haydjanggg6578
The boiling point of propanoic acid is higher than that of 1-butanol because: propanoic acid has a higher molecular weight than 1-butanol. propanoic acid is more soluble in water than 1-butanol. propanoic acid is a better hydrogen bond donor than 1-butanol. propanoic acid forms hydrogen bonded dimers and 1-butanol does not. 1-butanol forms hydrogen bonded dimers and propanoic acid does not.
Answers: 2
You know the right answer?
Which substance in this redox reaction is the oxidizing agent? cu + 2agno3 → 2ag + cu(no3)2 a. n b...
Questions in other subjects:


Social Studies, 02.08.2019 20:30

Mathematics, 02.08.2019 20:30


Chemistry, 02.08.2019 20:30



Mathematics, 02.08.2019 20:30

Advanced Placement (AP), 02.08.2019 20:30



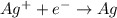
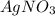 is considered as an oxidizing agent and therefore the correct answer is Option B.
is considered as an oxidizing agent and therefore the correct answer is Option B.