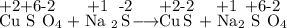Chemistry, 05.07.2019 09:00 DeGeneral8468
Did an oxidation-reduction reaction occur in the reaction between copper sulfate and sodium sulfide? cuso4 + na2s → cus + na2so4 a. this is a redox reaction because copper gained electrons. b. this is a redox reaction because sodium lost electrons. c. this is a redox reaction because sulfur gained electrons. d. this is not a redox reaction because no electrons were transferred. e. this is not a redox reaction because oxygen did not gain electrons. in which reaction is precipitation occurring? a. mgcl2(aq) + cuso4(aq) → cucl2(aq) + mgso4(aq) b. cdso4(aq) + k2s(aq) → cds(s) + k2so4(aq) c. naoh(aq) + nh4cl(aq) → nacl(aq) + nh4oh(aq) d. k2so4(aq) + naoh(aq) → k2oh(aq) + naso4(aq) e. hno3(aq) + koh(aq) → kno3(aq) + h2o(l)

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 16:30, lil3114
Imagine that you own a property that is exactly 2.2 acres large. you want to sell your property, but your realtor tells you that you cannot sell your land by the acre. in order to sell your land you need to determine the area you own in units of square meters? given that there are 1.6 kilometers in 1 mile and 640 acres in 1 square mile, what is the area of land that you own in square meters square meters?
Answers: 2

Chemistry, 22.06.2019 06:00, lindseyklewis1p56uvi
Ethanol (c2h5oh) is produced from the fermentation of sucrose in the presence of enzymes. c12h22o11(aq) + h2o(g) 4 c2h5oh(l) + 4 co2(g) determine the theoretical yield and the percent yields of ethanol if 680. g sucrose undergoes fermentation and 326.5 g ethanol is obtained. theoretical _ g _ percent %
Answers: 1

Chemistry, 22.06.2019 10:30, Riplilpeep
What is the empirical formula of c6h18o3? ch3o c2h5o c2h6o c2h5o5
Answers: 1
You know the right answer?
Did an oxidation-reduction reaction occur in the reaction between copper sulfate and sodium sulfide?...
Questions in other subjects:








History, 07.04.2020 22:38