
Chemistry, 05.07.2019 18:00 vapelordcarl69
Describe the relationship of the atoms shown by: a) the rearrangement of atoms is a chemical change. b) stability of the atoms in the molecule means a physical change. c) a change in state of matter from a liquid to gas is a physical change. d) a change in state of matter from solid to liquid is a physical change
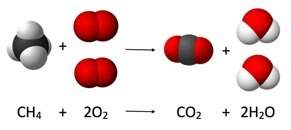

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 23.06.2019 00:00, scottykinkade7860
Mercury turns to a vapor at 629.88 k. how much heat is lost when 75.0 g of mercury vapor at 650 k condenses to a liquid at 297 k?
Answers: 1

Chemistry, 23.06.2019 04:20, vliu732
The reaction below shows a system in equilibrium. how would a decrease in temperature affect this reaction? a. the rate of formation of the gases would increase. b. the equilibrium of the reaction would shift to the left. c. the equilibrium would shift to cause the gases to sublime into solids. d. the chemicals on the left would quickly form the chemical on the right.
Answers: 1

Chemistry, 23.06.2019 09:00, blossie94681
The vapor pressure of water at 25.0°c is 23.8 torr. determine the mass of glucose (molar mass = 180 g/mol) needed to add to 500.0 g of water to change the vapor pressure to 22.8 torr.
Answers: 1
You know the right answer?
Describe the relationship of the atoms shown by: a) the rearrangement of atoms is a chemical change...
Questions in other subjects:


Mathematics, 07.04.2021 01:00


English, 07.04.2021 01:00

Geography, 07.04.2021 01:00





Mathematics, 07.04.2021 01:00



