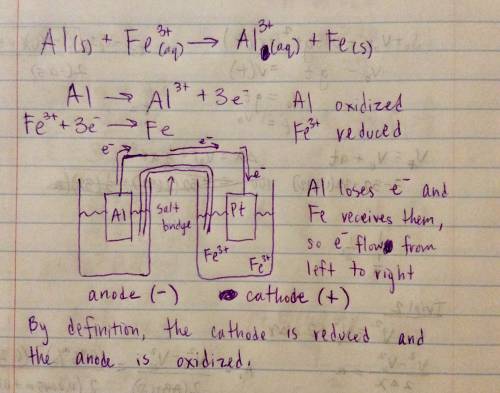
Galvanic cell runs on the following reaction: al (s) + fe3+ (aq) → al3+ (aq) + fe (s) draw a diagram for this galvanic cell, labeling the electron flow, the anode and cathode, and the positive and negative sides of the galvanic cell. answer only if you know show the diagram ! i don't understand

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 05:50, Makoshark6887
According to coulomb's law, how would the electrical force between particles change if the product of their electrical charge increased?
Answers: 1


You know the right answer?
Galvanic cell runs on the following reaction: al (s) + fe3+ (aq) → al3+ (aq) + fe (s) draw a diagra...
Questions in other subjects:

Mathematics, 13.12.2020 14:00

Arts, 13.12.2020 14:00

Biology, 13.12.2020 14:00

English, 13.12.2020 14:00

Spanish, 13.12.2020 14:00

Mathematics, 13.12.2020 14:00

Mathematics, 13.12.2020 14:00