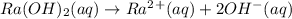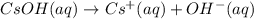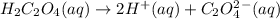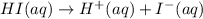Chemistry, 07.07.2019 04:00 clebby3056
Which it the compounds h2c2o4, ra(oh)2, csoh, and hi, behave as bases when they are dissolved in water?

Answers: 1


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 04:40, deedee363
*will mark you brainliest + 15 points ** why does the equilibrium of a system shift when the pressure is increased? a. to maximize the stress on the system b. to stop restoring equilibrium to the system c. to increase the total moles of gas in the system d. to decrease the total moles of gas in the system
Answers: 3


Chemistry, 22.06.2019 10:50, mi364
A100 kmol/h stream that is 97 mole% carbon tetrachloride (ccl4) and 3% carbon disulfide (cs2) is to be recovered from the bottom of a distillation column. the feed to the column is 16 mole% cs2 and 84% ccl4, and 2% of the ccl4 entering the column is contained in the overhead stream leaving the top of the column. calculate the mass and mole fractions of ccl4 in the overhead stream, and determine the molar flow rates of ccl4 and cs2 in the overhead and feed streams. 12. mw_ccla- 153.82; mw_cs2-76.14.
Answers: 3
You know the right answer?
Which it the compounds h2c2o4, ra(oh)2, csoh, and hi, behave as bases when they are dissolved in wat...
Questions in other subjects:

Mathematics, 16.09.2020 09:01

Mathematics, 16.09.2020 09:01

Mathematics, 16.09.2020 09:01

Mathematics, 16.09.2020 09:01

Biology, 16.09.2020 09:01

Mathematics, 16.09.2020 09:01

Mathematics, 16.09.2020 09:01

Mathematics, 16.09.2020 09:01

History, 16.09.2020 09:01

Mathematics, 16.09.2020 09:01

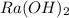 and CsOH .
and CsOH .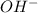 and so when dissolved in water, they give
and so when dissolved in water, they give 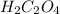 and HI gives
and HI gives 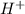 when dissolved in water and so these are acids.
when dissolved in water and so these are acids.